AMSE research team led by Prof. Hyung Koun Cho (Young-Been Kim Ph.D.) …
페이지 정보

본문

[Figure1] Researcher Young-Been Kim
AMSE Young-Been Kim under the Ph.D. program in Prof. Hyung Koun Cho‘s research team fabricated a high-efficient solar energy water splitting system that operates in alternating current (AC) voltage.
By designing an electrochemical deposition-based precursor synthesis process in order to micro-control based on thermodynamic morphology of Cu-In-Se (CIS), the research team developed a semiconductor structure where secondary phases coexist through the selenization process. Based on that, the team built the pulse-driven system in which enables high hydrogen evolution.
The multiphasic structure has improved the charge transport efficiency due to the expansion of the depletion region formed inside and on the surface of semiconductor electrodes. It can also generate photocurrent at reduction under negative (-) voltage and oxidation under positive (+) voltage due to the presence of multiphase. Consequently, the pulse-driven system is the new mechanism that can fully leverage the entire photocurrent generated out of AC output.
Pulse-driven photoelectrochemical (PEC) water splitting has been introduced to improve Hydrogen evolution efficiency by destroying the charge accumulation and electrical double layer (ionic layer) of the electrode surface causing the loss. Herein, the research team demonstrated first significant performance in the field of photoelectrochemical water-splitting employing semiconductor electrodes. Furthermore, efficient hydrogen evolution was confirmed by suppressing the formation of large-size cluster bubbles and facilitating hydrogen ion adsorption. Specifically, Through a 156% improvement in hydrogen evolution over the DC voltage system, Researcher Kim attested the superior performance of pulse-driven PEC water splitting system.
Along with public attention on hydrogen energy in which shows high efficiency as renewable energy to replace petroleum, water-splitting research using light to generate hydrogen is increasingly being active. Thus, research has been conducted on photoactive materials that can generate high current under sunlight. However, it was limited to single conductivity types in which shows very low efficiency of photo-generated charge transport due to the neutrality of the inner area of absorbers.
Consequently, the researchers, by focusing on the development of multi-phase chalcogenide materials that enhance charge transport efficiency, enabled utilizing both photocathode and photoanode by controlling the precursor synthesis and selenization process.
This research was supported by Samsung Research Funding &Incubation Center of Samsung Electronics [grant number SRFC-MA170206].
The paper was published online in January 2021 in Applied Catalogis B: Environmental (IF 16.683), a SCI journal within the top 1.94% in materials and environmental engineering category.
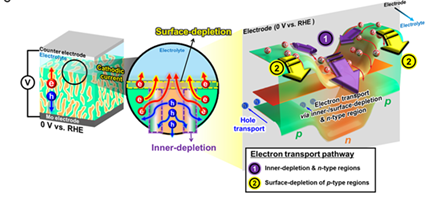
[Figure 2] The photo-generated minority carriers effectively flow into interleaved depletion regions and transport along the externally induced built-in field at 0 V vs RHE.
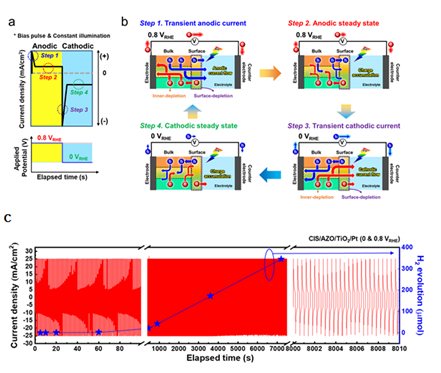
[Figure 3] (a) Sequential steps illustrating one-cycle of current behavior in chronoamperometry result under pulse bias of 0 and 0.8 V vs RHE under light illumination (b) schematic diagrams for charge transport mechanism under constant light illumination from p-n interleaved CIS photoelectrode, where (Steps 1 and 3 ) the high-density charge transport through interleaved depletion regions; and (Steps 2 and 4 ) charge accumulation at the surface region and equilibrium state indicating a saturated steady state. (c) CA results and hydrogen evolution rate of the CIS/AZO/TiO2/Pt photoelectrode at pulse bias of 0 and 0.8 V vs RHE with alternating frequency of 150 cpm (1 M Na2SO4 electrolyte buffered at pH 5 with potassium borate solution) under constant 1 sun light illumination.
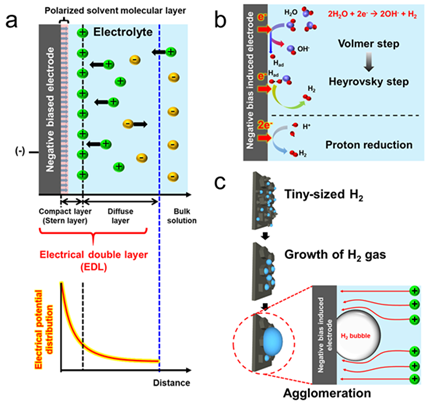
[Figure 4] (a) Electrical double layer (EDL) formation under negative biased electrode (b) Hydrogen generation mechanisms through the reduction reaction in the electrolyte (c) Active area loss due to gas agglomeration produced when DC power has driven
관련링크
- 이전글Prof. Jeong Min Baik’s Research Team Suggest a New Paradigm of Study on Thermoelectric Energy-harvesting 21.04.19
- 다음글Minsu Choi of AMSE wins a Silver award at the 27th HumanTech Paper Award 21.04.16
댓글목록

William님의 댓글
William 작성일
https://www.adidasyeezywebsite.us.org/ Adidas Yeezy 700
https://www.jordan1.us.org/ Nike Air Jordan 1
https://www.nikesnew.us.com/ Nikes
https://www.jordan4.us.org/ Jordan 4 Retro
https://www.newnikesshoes.us.org/ Nike Shoes
https://www.nikefree.us.org/ Nike Free Rn 2019
https://www.jordan27.us/ Jordan 27
https://www.nikeair-force1.us.org/ Nike Air Force 1
https://www.nikezoomshoes.us.com/ Nike Zoom
https://www.charmspandoras.us.com/ Pandora Charms
https://www.nikeairjordan.us.org/ Nike Air Jordan Shoes
https://www.nikeairmaxs-270.us.com/ Nike Air Max 270
https://www.nikeshoesstores.us.com/ Nike
https://www.airjordan33.us/ Air Jordan 33
https://www.nikerunningshoesforwomen.us.com/ Nike Running Shoes Women
https://www.jordan21.us/ Air Jordan 21
https://www.airmaxs.us.org/ Air Max 720
https://www.airjordan-retros.us/ Jordan Retros
https://www.nikemetcons.us.com/ Nike Free rn
https://www.jordan16.us/ Air Jordan 16
https://www.nikewholesalesuppliers.us.com/ Nike Wholesale Dealer
https://www.nikefactory-store.us.com/ Nike Outlet
https://www.jordans33.us/ Jordan 33
https://www.jordan18.us/ Jordan 18
https://www.foamposites.us.org/ Nike Air Foamposite
https://www.adidasstoreoutlet.us.com/ Adidas Outlet
https://www.huaracheshoes.us.com/ Huarache Shoes
https://www.airmax-outlet.us.com/ Air Max
https://www.nikesbdunk.us.com/ Nike SB
https://www.jordan15.us/ Air Jordan 15
https://www.pandorajewelrycz.us/ Pandora CZ Ring
https://www.jordan33.us.org/ Jordan 33 shoes
https://www.pandorajewelrycharmscanada.ca/ Pandora Earrings
https://www.jordan30.us/ Jordan 30
https://www.jordan22.us/ Air Jordan 22
https://www.jordans32.us/ Jordans 32
https://www.wholesalenikeshoesclothing.us.com/ Cheap Wholesale Nike Shoes And Clothing
https://www.retro12.us/ Retro 12
https://www.airjordanretro.us.org/ Jordan Shoes
https://www.yeezysboost350v2.us.org/ Yeezy Boost 350 V2
https://www.nikewholesale.us.org/ Nike Wholesale
https://www.jordanswholesale.us.org/ Wholesale Retro Jordans
https://www.cheapjordanswholesalefreeshipping.us/ Cheap Jordans
https://www.officialpandorajewelry.us/ Pandora Jewelry Official Site
https://www.jordan17.us/ Jordan 17
https://www.nikeepicreactuptempo.us.org/ Air Uptempo
https://www.nikeshops.us.com/ Nike Shoes
https://www.nikefoampositeacghyperdunk.us.com/ Nike Foamposite
https://www.pandorajewelryofficialsites.us/ Pandora Jewelry Official Site
https://www.charmspandora.us.org/ Pandora Charms
https://www.nike-outlets.us.com/ Nike Outlet
https://www.nflshoponline.ca/ NFL Shop
https://www.jordans14.us/ Jordan 14
https://www.michaeljordan-shoes.us/ Michael Jordan Shoes
https://www.nikeair-max95.us.com/ Nike Air Max 95
https://www.jordan24.us/ Jordan 24
https://www.jordan-12.us.org/ Air Jordan Retro 12
https://www.jordan20.us/ Air Jordan 20
https://www.pandorajewelryshops.us.com/ Pandora Jewelry 70 Off Clearance
https://www.cheapshoeswholesalefromchina.us/ Cheap Nike Shoes From China Wholesale
https://www.nikecortezshox.us.org/ Nike Shox Women
https://www.nikeairforces.us.com/ Nike Air Force Ones
https://www.redbottomslouboutinshoes.us.org/ Red Bottom Heels
https://www.wholesaleshoessneakers.us/ Wholesale Adidas
https://www.jordan29.us/ Air Jordan 29
https://www.nikeshoessale.us.org/ Nike Shoes
https://www.nikeoutletstoreonlines.us.org/ Nike Outlet Store
https://www.pandora-jewelrysite.us/ Pandora Jewelry
https://www.ringspandora.us/ Pandora Ring
https://www.airjordans11retro.us/ Air Jordan 11 Retro
https://www.jordan6s.us/ Jordans 6
https://www.jordans34.us/ Jordan 34
https://www.mlbjerseysshop.ca/ MLB Jerseys
https://www.jordan35.us/ Jordan 35
https://www.wholesaleshoesclothing.us/ Wholesale Shoes And Clothing
https://www.christianslouboutin.us.org/ Christian Louboutin
https://www.jordans28.us/ Jordan 28
https://www.newnikesneakers.us.org/ Nike Sneakers
https://www.nikesneakerss.us.com/ Nike Store
https://www.cheapjordansshoessale.us/ Cheap Jordans
https://www.jordans23.us/ Air Jordan 23
https://www.pandoraa.us/ Pandora
https://www.pandora-charmssale.us/ Pandora Charms
https://www.air-maxs.us.com/ Air Max
https://www.nikeairmaxs.us.org/ Air Max
https://www.jordans13shoes.us/ Jordan 13s
https://www.pandorasjewelrycharms.us/ Pandora Charms
https://www.jordans12.us/ Jordans 12
https://www.nbastorecanada.ca/ Raptors Jersey
https://www.jordan19.us/ Jordan 19
https://www.jordan11concordshoes.us/ Jordan 11 Concord
https://www.nmdr1.us.com/ Adidas NMD
https://www.nikeairmax270s.us.com/ Nike Air Max 270
https://www.pandorajewelryauthentic.us/ Pandora
https://www.nikewomensshoes.us.com/ Nike Women's Shoes
https://www.airmaxs97.us.com/ Nike Air Max 97 Ultra
https://www.christianlouboutinshoess.us.com/ Louboutin Shoes
https://www.air-max2019.us.org/ Nike Air Max 2019
https://www.pandorasbracelets.us/ Pandora Bracelet Charms
https://www.jewelryspandora.us.com/ Pandora Charms
https://www.jordan-aj1.us/ Jordan AJ 1
https://www.wholesalejordansfactory.us/ Wholesale Jordans From China Factory
https://www.nikesoutletfactory.us.com/ Nike Outlet
https://www.jordan2s.us/ Jordan 2s
https://www.nikerosheblazers.us.com/ Nike Blazers
https://www.airmax720.us.org/ Nike Air Max 720
https://www.nikeairforceones.us.org/ Air Force Ones
https://www.nikeoutlet-store.us.org/ Nike Outlet
https://www.nikesoutletstores.us.com/ Nike Outlet
https://www.nikerunningshoes.us.org/ Nike Running Shoes
https://www.airforce1s.us.org/ Air Force 1 Shoes
https://www.jordan25.us/ Jordan 25
https://www.nikeshoesoutletstoreonlineshopping.us.com/ Nike Factory Outlet Store Online
https://www.pandorabracelet-clearance.us.com/ Pandora Bracelets
https://www.pandora-jewelry-charms.us/ Pandora Jewelry Charms
https://www.pandorajewelryblackfriday.us.com/ Pandora Charms
https://www.jordan5whatthe.us/ What The Jordan 5
https://www.nikejordan1.us.com/ Nike Jordan 1 Low
https://www.cheapshoeswholesalefreeshipping.us/ Cheap Nikes From China
https://www.nikeairhuaraches.us.com/ Nike Air Huarache
https://www.wholesaleadidas.us.com/ Wholesale Adidas Sneakers
https://www.lebronsjamesshoes.us.com/ Lebron James Shoes
https://www.officialpandorarings.us/ Pandora Rings
https://www.jordan31.us/ Jordans 31
https://www.huaraches.us.org/ Huarache
https://www.nikeairforce1s.us.org/ Nike Air Force 1
https://www.cheapjordanshoessuppliers.us.org/ Cheap Jordan 12
https://www.toddlerbabyinfantjordans.us/ Baby Jordan
https://www.cheapjordansshoeswholesale.us.org/ Cheap Jordans Wholesale
https://www.nike-clearance.us.org/ Nike Clearance
https://www.pandoraoutletsjewelry.us.com/ Pandora Outlet
https://www.newjordans.us.org/ New Jordan
https://www.nikes.us.org/ Nike Shoes
https://www.airjordans13.us/ Air Jordan 13 Retro
https://www.jordanshoess.us.org/ Jordan Shoes
https://www.shoeswholesalesuppliers.us/ Nike Wholesale
https://www.nikecanadashoesshop.ca/ Nike Shoes
https://www.cheapadidasshoes.us.org/ Adidas Shoes
https://www.jordan26.us/ Jordan 26
https://www.canadashoesoutlet.ca/ Nike Shoes
https://www.diorjordans.us/ Dior Jordans
https://www.nikeshoesdeals.us.com/ Nike Shoes
https://www.nikeairzoom.us.com/ Nike Zoom
https://www.jordan11s.us.org/ Jordan 11s
https://www.nhlshops.ca/ NHL Jerseys
https://www.nikeoutletshoes.us.org/ Nike Outlet
https://www.nikesoutletstore.us.com/ Nike Outlet Store Online Shopping
https://www.nikess.us.com/ Nikes
https://www.nikeblackfridaycybermonday.us.org/ Nike Black Friday Deals 2019
https://www.nikeshoesformens.us.com/ Nike Shoes Men
https://www.nhljerseysstore.ca/ NHL Jerseys
https://www.kidsjordans.us/ Kids Jordans
https://www.nikerosheblazer.us.org/ Nike Roshe Men
https://www.jerseysstore.ca/ NBA Jerseys
https://www.wholesaleshoescheap.us/ Cheap Adidas Shoes
https://www.shoesstores.ca/ Nike Canada Store
https://www.jordan11lowretro.us/ Air Jordan 11 Low
https://www.nikeoffwhite.us.org/ Nike x Off White
https://www.cheapjerseyswholesale.ca/ Cheap Jerseys Wholesale
https://www.pandorajewelryoutlets.us.com/ Pandora Jewelry Outlet

Lisa님의 댓글
Lisa 작성일Fill up your travel map with the help of https://www.thingstodo-near.me - seize the opportunity to discover the world around you.

Yeezy V2 Outlet님의 댓글
Yeezy V2 Outlet 작성일
https://www.yeezyv2-outlet.us Yeezy Slide
https://www.yeezyofficialwebsites.org Yeezy Official Websites
https://www.yeezyofficialwebsites.org Adidas Yeezy Outlet
https://www.yeezyofficialwebsites.org Yeezy Supplys
https://www.yeezyofficialwebsites.org Yeezy Shoes
https://www.yeezysslide.us Yeezy Slides
https://www.yeezysslide.us Yeezy V2
https://www.yeezyv2-outlet.us Yeezy Supply
https://www.yeezyv2-outlet.us Yeezy Outlet
https://www.yeezyv2-outlet.us Yeezy V2 Outlet
https://www.yeezysboost.us Yeezy Supplys
https://www.yeezysboost.us Yeezy Boost
https://www.yeezysboost.us Yeezys Boost
https://www.yeezysboost.us/sitemap.xml
https://www.yeezyv2-outlet.us/sitemap.xml
https://www.yeezysslide.us/sitemap.xml
https://www.yeezyofficialwebsites.org/sitemap.xml
Tags:yeezy shoes, Yeezy, Adidas Yeezy,yeezy supply, yeezy slide


